Table of Contents
What is Restasis [R]?
Restasis is 0.05% cyclosporin that was developed in 2003 by Allergan in the United States. Cyclosporin had originally been using to help minimize an elevated immune response post organ transplant.
It became the first FDA approved drop to treat moderate to severe dry eye.
The main goal of a topical application of cyclosporine is to act as an immunosuppressant and reduce inflammation to the surface as well.
Clinical Application [R]
This medication is best suited for patients with moderate to severe forms of dry eye. This includes ocular staining scores, tear production, and fluctuating visual symptoms.
There have been no studies to determine the efficacy of Restasis for dry eye treatment in the pediatric (<18 years old), and geriatric (>65-year-old) population.
Caution should be taken for patients who are pregnant, or breast feeding. With cyclosporine medication there is a high correlation between the medication and maternal toxicity. Oral dosing of cyclosporine has also been found in breast milk, but there have been no topical studies to determine this.
It’s probably in your patient’s best interest if they are planning to bear children or are pregnant, this is not the medication to be using
Special note should also be taken for patients who have had a history of herpes keratitis as there have been no official studies that document using this drug after recovery.
Contraindications [R]
Any patients who have shown hypersensitivity to Restasis, or the ingredients from the drop should avoid being used this.
Any patients who have signs of ocular disease should also not use this, as there is a localized immune-suppressive effect (more on this later).
Mechanism of Action [R]
Restasis is topically applied and is classified as immunomodulatory because it can reduce the immune system response without allowing opportunistic infection to occur.
The drug functions to inhibit pro-inflammatory cytokines on the surface of the eye and inhibits T cell activation as well BUT still allowing the next step of the immune response to take place. This permits an appropriate immune response when faced with infection.
Also, there are some findings that show it up-regulates TH2-anti-inflammatory cytokines (which help to minimize inflammation on the ocular surface).
Systemic Absorption [R]
Topical application of Restasis surprisingly has VERY little systemic absorption
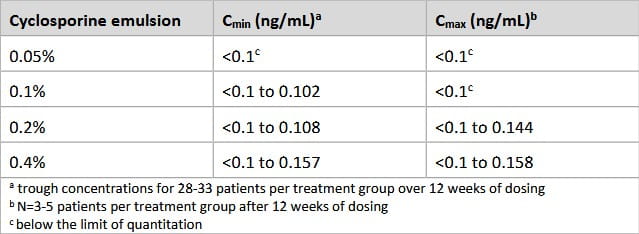
This graph shows a concentration of 0.05% cyclosporine has negligible impact on serum concentration levels.
Increasing the concentration to 0.4% cyclosporine (which would be higher than CEQUA right now) cant even crack the level of 1ng/mL
Compared to an oral / IV dosage of cyclosporine which can elevate the blood serum levels up to 250ng/mL for up to one week after dosage, and 100-250ng/mL 6-12 months after performing an organ transplant [R].
Storage
Ideally it should be stored at room temperature, but there are some studies that show placing it into the fridge helps to minimize ocular surface burning during installation.
Dosing
The current recommended dosing schedule is BID, but there has been some studies that have shown that higher frequency dosing can also help to improve corneal staining as well [R]
Further studies would need to be performed at even higher frequency installations to determine efficacy / safety profile.
Side effects
The most common ADR is burning upon installation
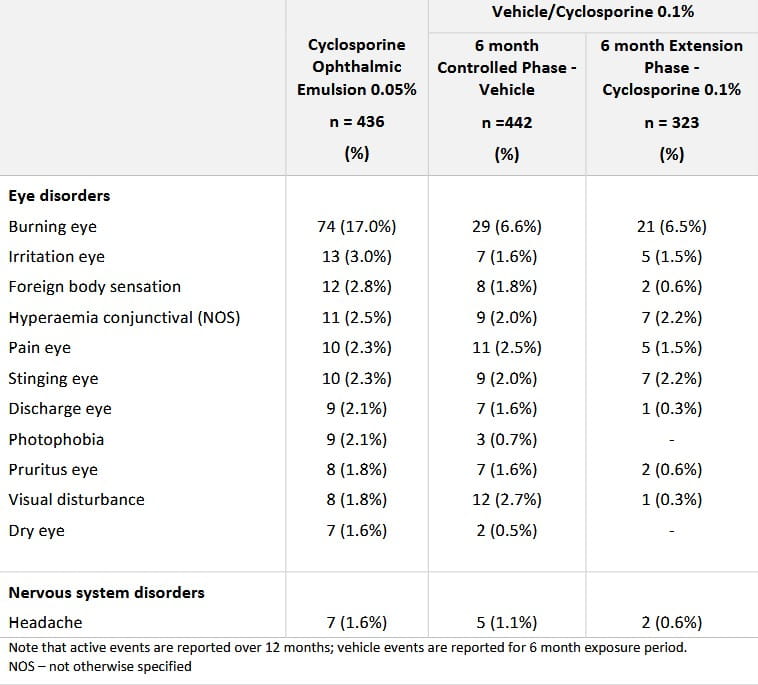
No serious adverse reactions were reported during clinical trials
Post market adverse drug reactions
The ADRs once the drug hit the market was very similar to clinical trials – burning upon installation. However, there were some additional ADRs not reported before such as; ocular injuries to the cornea / lid or face swelling / severe angioedema
Clinical trial results [R]
The results with BID Restasis dosing are very impressive
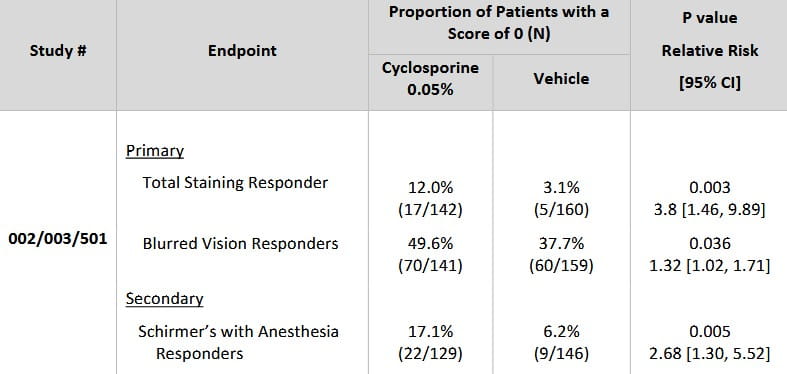
Total Staining
After 6 months of BID dosing, staining scores improved from a base line of 12% when compared to vehicle delivery.
Blurred vision
Subjective reports also reported an improvement when compared to the 6-month baseline. Approximately 50% of patients had a positive response compared to only 38% of vehicle delivery.
Schirmer with anesthesia
A statistically larger number of patients (17%), treated with Restasis improved their Schirmer score when compared to vehicle (6%)
Sjogren’s vs non-Sjogren’s
0.05% cyclosporine treatment was most efficacious in treating Sjogren’s ocular staining scores when compared to exclusively vehicular treatment.
My Restasis Review
I’ve placed a couple of my patients on Restasis so far with really good results.
Most notably, some of my Sjogren’s patients have really appreciated starting the medication and have also shown great results in their subjective symptoms as well.
Steroid loading dose
A little off label, but I have also talked with some colleagues about using Lotemax BID for 1 month simultaneously with Restasis to provide a pseudo-loading dose to reduce ocular inflammation, and reduce burning / stinging,
The studies also seem to help support that claim as well [R].
[yop_poll id=”3″]